TRePEV
The valence (shell) electron pair repulsion theory (TRePEV or TRPECV), VSEPR theory , or Gillespie theory is a model used in chemistry to predict the shape of polyatomic molecules or ions. It is based on the degree of electrostatic repulsion of valence electron pairs around the atom. It is also called the Gillespie-Nyholm theory after its two main developers.
The premise of TRePEV is that the valence electron pairs around an atom repel each other, and therefore, adopt a spatial arrangement that minimizes this repulsion, determining the molecular geometry. The number of valence electron pairs around an atom, both bonding and nonbonding, is called the steric number.
TRePEV is generally compared and contrasted with valence bond theory, which deals with the molecular shape through orbitals that are energetically accessible to form bonds. Valence bond theory deals with the formation of sigma and pi bonds. Molecular orbital theory is another model for understanding how atoms and electrons assemble into molecules and polyatomic ions through the formation of molecular orbitals.
TRePEV has been criticized for not being quantitative, and therefore limited to qualitatively obtaining the molecular geometries of molecules and covalent polyatomic ions, despite the fact that it is structurally precise. However, force fields have also been developed in molecular mechanics based on TRePEV.
History
The idea of establishing a molecular correlation between the geometry of a molecule and the number of valence electrons was first presented in 1940 by Nevil Sidgwick and Herbert Powell at the University of Oxford. In 1957 Ronald Gillespie and Ronald Sydney Nyholm of University College London refined the concept by building a detailed theory that allowed choosing, among various geometric alternatives, the most suitable for a given molecule.
Description
Valence Electron Pair Repulsion Theory (VERT) is based on the idea that the geometry of a molecule or polyatomic ion of type ABn, where A is the central atom and B the peripheral atoms or ligands, is mainly conditioned by the Coulomb-like repulsion between the pairs of electrons in the valence shell around the central atom.
The predicted geometry is that which gives the pairs of electrons in the valence shell the minimum energy. In fact, it just so happens that when an electron distribution is just right, it coincides with a minimal interelectron repulsion.
The pairs of electrons can be of two types depending on whether or not they are part of a bond, being classified as bonding pairs and loose pairs, (also called free pairs, or non-bonding pairs).
There are three types of repulsive interactions between the pairs of electrons in a molecule, each with a certain value of intensity. Ordered from highest to lowest repulsion, the possible interactions are:
- The repulsion pair non-linking - pair non-linking (PNE-PNE).
- The repulsion pair not enlazante - pair enlazante (PNE-PE).
- The repulsion pair enlazante - pair enlazante (PE-PE).
Given this division into two classes of pairs, any such molecule can be expressed as ABnEm, where n is the number of binding pairs and m that of non-bonding pairs.
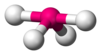
A molecule with a central atom that satisfies the octet rule will have four pairs of electrons in its valence shell. If all four pairs are bonding, the bonded atoms will be arranged at the vertices of a regular tetrahedron. The tetrahedral bond angle is 109.5°.
As mentioned, the repulsion nonbonding pair - nonbonding pair (PNE-PNE) is considered stronger than the repulsion nonbonding pair - bonding pair (PNE-PE), which is in turn stronger than the repulsion bonding pair - bonding pair (PE-PE). Then, the angle formed by two bonding pairs will be smaller than the one formed by the pairs (PNE-PE) and this in turn smaller than the one formed by the pairs (PNE-PNE ).
In this sense it agrees quite well with the experimental data. The explanation to justify a greater intensity in the PNE-PNE interaction, and therefore a greater opening angle than in the other interactions, is based on the greater dispersion of the electron cloud of the electrons housed in nonbonding orbitals.
Additional rules for the prediction of molecular geometry
- The repulsions exercised by the bond pairs decrease by increasing the electronivity of the peripheral atoms. For example, if compared NH3 with him NF3molecules with the central atom belonging to the same group, and same geometry, (piramidal), the focal angles are respectively 106.6o and 102.2o. Please note that the fluoride has an electronegativity of 3.98, on the Pauling scale and hydrogen of only 2.20, compared to nitrogen with 3.
- The formation of multiple (double, triple) links does not affect the stereometry of the molecule, which is mainly determined by the links σ σ {displaystyle sigma } and the pairs of free electrons.
- The repulsion between pairs of non-linking electrons of atoms with full layers is greater than the repulsion between pairs of non-linking electrons belonging to atoms with layers of incomplete valence, as can be seen in the progressive decrease of angles in the sequence of molecules H2S, H2Se, H2Te, H2O.
- When the central atom presents the incomplete layer of valence, and one or more empty free orbitals, there is a tendency for the pairs of electrons free from the peripheral atoms to transfer to the first, as is the case in the BF3. Free electron pairs that surround fluoride atoms can yield to the orbital pz Boron vacuum.
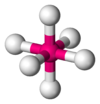
- When there are 5 or 7 pairs of electrons the positions are not completely equivalent. For example, in a molecule with wheat bipyramid geometry (5 pairs), as is the case of PF5, you can distinguish axial positions and equatorial positions.
REPEV versus other theories
TRePEV is compared and contrasted with valence shell bonding theory, which determines the shape of molecular geometry through orbitals that are energetically accessible for bonding. Molecular orbital theory concentrates more on the formation of sigma and pi bonds. Molecular orbital theory is a more sophisticated model for understanding how atoms and electrons assemble into polyatomic molecules and ions.
The Ax method is commonly used to find the geometry of molecules following the TRePEV theory.
Examples
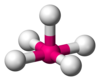
Methane (CH4) is tetrahedral because there are four pairs of electrons. The four hydrogen atoms are positioned at the vertices of a tetrahedron, and the bond angle is 109.5°. It is a molecule of the AB4 type. A is the central atom and B represents the other atoms.
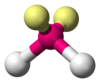
Ammonia (NH3) has three pairs of electrons used in bonds, but there is one lone pair of electrons on the nitrogen atom. It is not bound to any other atom, but still influences geometry through repulsions. As in methane, there are four regions of electron density. Therefore, the general orientation of the regions of electron density is tetrahedral. Then again, there are only three outer atoms. It is an AB3E type molecule because the loose pair of electrons is represented by an E. The general shape of the molecule is a trigonal pyramid because the loose pair is not "visible". The geometry of a molecule is obtained from the relationship of the atoms even though it can be influenced by the lone pairs of electrons.
Due to the greater repulsion exerted by the non-bonding pair of electrons of N, while the predicted HCH bond angle is 109.5º, the HNH of ammonia should be less.
Exceptions
There are groups of compounds where TRePEV predictions fail.
Transition metal compounds
Many structures of transition metal compounds cannot be correctly described, which can be attributed to the interaction of the d electrons of the inner electron shell with the ligands, which lie beyond of the coordination sphere of lone pairs.
Contenido relacionado
Gas chromatography
Molecularity
Glass