Major histocompatibility complex
The major histocompatibility complex or MHC (also MHC for ccomplex pmajor histocompatibility or MHC, an acronym for mmajor histocompatibility complex), is a family of genes found in all vertebrates and located on the short arm of chromosome 6 in humans, whose function is to encode molecules (glycoproteins) called human leukocyte antigens or histocompatibility antigens, which participate in the presentation of antigens to T lymphocytes allowing the activation of critical processes in the generation of the immune response. In general, the CMH makes it possible to distinguish what is self from what is foreign.
In humans, MHC was first called the HLA complex (from the English acronym for human leukocytic antigen), because MHC molecules were discovered as antigens that differentiate leukocytes from different people and that produce the recipient's immune response in transplants, producing rejection.
The MHC is the most variable area of the human genome and contains numerous functional genes, characterized by large polymorphisms. The products of these genes are also mentioned by many authors as MHC or HLA.
History
HCM was discovered in the 1950s. Previously, in the 1900s the field of immunogenetics was opened up with the discovery of blood groups, followed by the discovery of the Rh factor in 1940.
In 1936, Peter Gorer was the first to describe a histocompatibility complex in mice. His research was followed by George Snell who suggested that the rejection of tissues by the mouse was due to incompatibility of some antigens. This MHC was named "H-2" (after the antigen 2 described by Gorer) and is similar to MHC from several species.
In 1952, Jean Dusset hypothesized that a histocompatibility system similar to that of red blood cells must be present in white blood cells. He was able to demonstrate it with the agglutination of leukocytes in the serum of a polytransfused patient. His work in this regard earned him the Nobel Prize in 1958.
The first specific identification of a CMH product was made in 1958, through the study of reactive serum. The MAC antigen, corresponding to the current HLA-A2, was identified. In the 1960s, the polymorphism was confirmed with the work of Jon van Rod, Rose Payne, and Walter Bodmer, who described the 4a and 4b (Bw4 and Bw6) antigens, and HLA-A2 and HLA-A3 in a study of women. multiparous.
In 1964, an international effort began to characterize the complex, called the International Histocompatibility Workshop (IHW). The area of chromosome 6 where HLA A, B and C are coded was then defined, which at that time was believed to be only expressed by leukocytes (hence the name HLA: human leukocytic antigen)); the complement system was also mapped as coming from the same zone. In the 1970s, HLA class IIs were identified and then, with advances in molecular biology, research focused on the gene level rather than its products.
In 1967, the term haplotype was used for the first time in relation to the CMH. In 1969, Baruch Benacerraf demonstrated that MHC was not only the cause of alloreactivity, but also enabled the activation of the immune response against a particular antigen. This discovery earned him the Nobel Prize in Medicine in 1980.
In 1987, Bjorkman succeeded in elucidating the structure of HLA-A2 and also of class II molecules. The nucleotide sequence of the MHC was defined in 1999 through the combined efforts of the MHC Sequencing Consortium under the direction of Stephen Beck, Daniel Geraghty, Hidetoshi Inoko, and Lee Rowen.
Between the years 1980 and 2000, knowledge of the alleles present in the MHC went from a few dozen, with several million possible allotypic combinations, to around 15,000 alleles. The HLA-B region became the most polymorphic genetic region of the human genome, followed by the HLA-A region. The MHC gene region contains extremely high gene density and diversity; Genetic variation within this region plays a vital role in susceptibility to autoimmune, infectious, and other diseases.
Organization of MHC genes and regions
Comparative analysis of the organization of the MHC region between widely separated species has revealed the presence of lineage-specific reorganizations within the region and changes in gene complexity.
The MHC region is the most gene-dense and polymorphic region of the mammalian genome, critical for immunity and reproductive success.
In non-human species
The structure of the MHC region is known in at least seven species of eutherian (placental) mammals, two birds, five teleost fish, and sharks. There are large differences in the organization of the MHC region between eutherian and non-mammalian mammals. In eutherians, the region is arranged along the chromosome in class I-II-III regions, is very gene-dense, and occupies a large area. In non-mammals, the MHC region generally contains fewer genes and the Class I and II regions are adjacent, except for teleosts, where the two regions are not linked. Of the fully sequenced MHC regions, the least complex is that of the chicken, which contains only 19 genes in 92 kb.
The MHC region in the marsupials Monodelphis domestica (short-tailed gray Didelphimorphia) is flanked by the genetic markers MOG and COL11A2, comprises 3.95 Mb and contains 114 genes, 87 shared with humans.
In humans
In humans, the MHC is made up of at least 4,000,000 and could be as many as 7,000,000 base pairs encoding more than 200 genes located on the short arm of chromosome 6.
The MHC is divided into 2 gene regions: the one closest to the centromere contains the class II genes, with the HLA–DRA, DRB, DQA, DQB, DPA, DPB, DNA, DMA, DMB and DOB loci; the region closest to the telomere contains the class I genes, with the HLA-A, B and C loci; between the two are the genes of some proteins of the complement system and others related to the immune response.
The region of the short arm of chromosome 6 that contains the MHC genes, has the information of:
- certain glucoproteins of the plasma membrane involved in antigen filing and processing mechanisms to T lymphocytes: they are grouped into class II genes (which encode class II CMH proteins) and class I genes (which encode class I CMH proteins)
- cytokines and supplemental system proteins, important in the immune response, but have nothing to do with the genes of CMH; these genes are grouped into class III.
Both types of molecules participate in the immune response, which allows the identification of self molecules and foreign ones (invaders), to eliminate the latter through different mechanisms.
Class I CMH
In eutherians, the MHC class I region contains a set of metopic genes whose presence and order is conserved between species. These molecules are expressed in all human cells, except red blood cells, germ cells, cells of pre-implantation embryos, and syncytiotrophoblast (embryonic tissue, not present in extrauterine life). Some cells, such as neurons, monocytes, and hepatocytes have low levels of MHC class I molecules (less than 103 per cell).
The MHC class I genes are located in the most distal zone (near the telomere) and are HLA-A, HLA-B and HLA-C. They are highly polymorphic: about 3,000 alleles have been described for each. Along with these "classic" (or 1a) genes are others called "non-classical" (or 1b): HLA-E, HLA-F, HLA-G, HFE and MIC (acronym for chains related to MHC). of class I, in English, MHC class I Chain-related): MICA and MICB. Except for the MICs, all of these genes encode glycoproteins, codominantly expressed as heterodimers in the membrane of each nucleated cell, non-covalently linked to the β2 microglobulin chain encoded by a gene on chromosome 15.
Its products have an immunoglobulin structure: they have an α-type heavy chain that is subdivided into three regions: α1, α2 and α3. These three regions are exposed to the extracellular space and are linked to the cell membrane via a transmembrane region. The α chain is always associated with a β2 microglobulin molecule, which is encoded by a separate region on chromosome 15.
The main function of class I gene products is the presentation of intracellular antigenic peptides to cytotoxic T lymphocytes (CD8+). The antigenic peptide is housed in a cleft formed between the α1 and α2 regions of the heavy chain, while the recognition of MHC-I by the cytotoxic T lymphocyte is done in the α3 chain. In this cleft formed by the α1 and α2 regions, peptides of between 8 and 11 amino acids are presented, which is why the presentation of the antigenic peptide must go through a fragmentation process within the cell that expresses it.
In humans, there are many isotypes (different genes) of Class-I molecules, which can be grouped into:
- "classic", whose function is to present antigens to T CD8 lymphocytes: within this group we have HLA-A, HLA-B and HLA-C.
- "not classical" (also called MHC class IB), with specialized functions: they do not present antigens to T lymphocytes, but they join NK cell inhibitor receptors; within this group are HLA-E, HLA-F, HLA-G. Therefore HLA-G proteins are called immunosuppressants and are expressed in the cytophoblast of the fetus. This expression is thought to prevent the fetus from being rejected as a transplant [1].
Class II MHC
MHC class II genes are located in the region closest to the centromere, and encode the two chains of the functional heterodimer: HLA-DR, HLA-DQ, HLA-DP, HLA-DM, and HLA-DO. Initially they were called Ir genes because they are involved in the immune response (from the English acronym for immune response).
These genes encode glycoproteins with an immunoglobulin structure, but in this case the functional complex is made up of two chains, one α and one β (each with two domains, α1 and α2, β1 and β2). Each of the chains is attached to the membrane by a transmembrane region, and both chains face each other, with contiguous domains 1 and 2, on the outside of the cell.
These molecules are expressed in a more restricted way than those of Class I in antigen-presenting cells, such as dendritic cells, macrophages, Langerhans cells, Kupffer cells, and B lymphocytes.
Antigen-presenting cells present extracellular antigenic peptides to helper T cells (CD4+). The antigenic peptide is housed in a cleft formed by the α1 and β1 domains, while the recognition of MHC-II by the helper T lymphocyte is done in the β2 chain. In this cleft formed by the α1 and β1 regions, peptides of between 12 and 16 amino acids are presented.
MHC class II molecules in humans present 5-6 isotypes, and can be grouped into:
- "classic", which present peptides to T CD4 lymphocytes; within this group we have HLA-DP, HLA-DQ, HLA-DR;
- "not classical", accessory, with intracellular functions (they are not exposed in the cell membrane, but in internal membranes, of the lysosomes); normally, they load antigenic peptides on the classical MHC-II molecules; in this group are included HLA-DM and HLA-DO.
In addition to class II MHC molecules, the Class-II region contains genes that encode antigen processing molecules, such as TAP (for transporter associated with antigen processing) and Tapasin.
Intermediate Region
Between the regions occupied by class I and class II genes, there are numerous genes that are important in the immune response but are not related to MHC. They encode several proteins that play immune functions: components of the complement system (such as C2, C3, and factor B) and molecules related to inflammation (cytokines such as TNF-α, Lymphotoxins A and B) or heat shock proteins.
MHC polymorphism
The MHC region has two characteristics that make it difficult for pathogens to selectively evade the immune response: it is polygenic and polymorphic, that is, it contains several different genes from class I and class II, and of each there are multiple variants or alleles.
MHC genes are expressed in a codominant manner. This means that the alleles inherited from both parents are expressed in an equivalent way:
- As there are three pairs of genes that encode the class I α chain, called in humans HLA-A, HLA-B and HLA-C, and each person inherits a game of each parent, any cell of an individual may express up to six different types of class I CMH molecules.
- Similarly, in the loci class II, each individual inherits a pair of HLA-DP genes (each with DPA and DPB, which encode α and β respectively), a pair of HLA-DQ genes (DQA and DQB, for α and β strings), a HLA-DRα gene (DRA1) and one or two HLA-DRβ genes (DRB1 and DRB3, -4 or -5) Thus, a heterozygous individual may inherit 6 or 8 alleles of Class-II, three or four of each parent.
The set of alleles present on each chromosome is called the MHC haplotype. In humans, each HLA allele is given a number. For example, for a given individual, their haplotype may be HLA-A2, HLA-B5, HLA-DR3, etc. Each heterozygous individual will have two MHC haplotypes, one on each chromosome (one of paternal origin and one of maternal origin).
MHC genes are highly polymorphic, which means that many different alleles exist in different individuals in the population. The polymorphism is so great that in a mixed (non-inbred) population no two individuals have exactly the same set of genes and MHC molecules, except for identical twins.
The polymorphic regions of each allele are found in the zone of contact with the peptide that is going to be presented to the lymphocyte. For this reason, the contact zone of each allele of the MHC molecule is highly variable, since the polymorphic residues of the MHC form specific clefts in which only a certain type of peptide residue can enter, which imposes a binding mode. very precise relationship between the peptide and the MHC molecule. This implies that each MHC molecule variant will be able to specifically bind only those peptides that fit properly into the MHC molecule's cleft, which is variable for each allele. Thus, MHC molecules have a broad specificity for peptide binding, since each MHC molecule can bind many, but not all possible types of peptides. This is an essential feature of MHC molecules: in a given individual, a few different molecules are enough to be able to present a vast variety of peptides.
On the other hand, within a population, the existence of multiple alleles ensures that there will always be some individual that possesses an MHC molecule capable of carrying the appropriate peptide to recognize a specific microbe.Evolution The MHC polymorphism ensures that a population will be able to defend itself against the enormous diversity of existing microbes, and that it will not succumb to the presence of a new pathogen or a mutated pathogen, because at least some individuals will be able to mount an adequate immune response. to defeat the pathogen. Variations in MHC sequences (responsible for polymorphism) result from the inheritance of different MHC molecules, and are not induced by recombination, as occurs with antigen receptors.
CMH Products
Functions of MHC Class I and Class II Molecules
Both types of molecules present antigenic peptides to T lymphocytes, responsible for the specific immune response to eliminate the pathogen responsible for the production of said antigens. However, MHC Class-I and II molecules correspond to two different pathways of antigen processing, and are associated with two different immune defense systems:
| Feature | Class II CMH | Class I CMH |
|---|---|---|
| Composition of stable peptide-CMH complex | Polymorphic chains α and β, peptide attached to both | Polymorphic chain α and microglobulin β2, peptide attached to α chain |
| Types of antigen-representative cells (APC) | Dendritic cells, mononuclear phagocytes, B lymphocytes, some endothelial cells, thymus epithelium | Almost all nucleated cells |
| T lymphocytes able to respond | Co-operative T lymphocytes (CD4+) | Cytotoxic T lymphocytes (CD8+) |
| Origin of antigenic proteins | Proteins present in endosomas or lisosomes (mostly internalized from the extracellular environment) | Cytolic proteins (mostly synthesized by the cell; they can also enter from the outside through phgosomas) |
| Enzymes responsible for the generation of peptides | Proteas of endosomas and lysosomes (such as catepsine) | Cosmetic proteasome |
| Peptide loading site on the CMH molecule | Specialized vesicular compartment | Endoplasmic revolving |
| Molecula involved in the transport of peptides and load on the molecules of the CMH | Invariant chain, DM | TAP (transport associated with antigen processing) |
T lymphocytes from a specific individual have a property called MHC restriction: they can only detect an antigen if it is presented by a MHC molecule from the same individual. This is because each T cell has a dual specificity: the T cell receptor (termed TCR for T cell receptor) recognizes some peptide residues and simultaneously some T cell residues. the MHC molecule presenting it. This property is very important in organ transplantation, and implies that, during their development, T lymphocytes must "learn" to recognize the individual's own MHC molecules, through the complex process of maturation and selection that takes place in the thymus.
MCH molecules can only present peptides, which implies that T lymphocytes, since they can only recognize an antigen if it is associated with a MHC molecule, can only react to antigens of origin protein (from microbes) and not to other types of chemical compounds (or lipids, or nucleic acids, or sugars). In contrast, the CD1 molecule, similar in structure to class I MHC molecules (3 α chains and one β-microglobulin), presents non-peptide antigens, usually lipids, to T lymphocytes. Each MHC molecule can present a single peptide each time, since the slit of the molecule only has space to accommodate one peptide. However, a given MHC molecule has a broad specificity, because it can present many (but not all) different peptides.
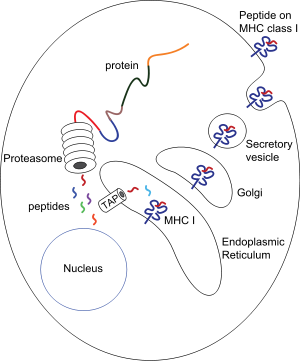
MHC molecules acquire the peptide that they present on the outside of the cell membrane during their own biosynthesis, inside the cell. Thus, the peptides presented by MHC molecules come from microbes inside cells, and this is the reason why T-lymphocytes, which only identify peptides when associated with MHC molecules, only detect microbes associated with MHC molecules. cells and trigger an immune response against intracellular microbes.
It is important to highlight that MHC class I molecules acquire peptides from cytosolic proteins, while MHC class II molecules acquire peptides from proteins contained in intracellular vesicles. Therefore, MHC class I molecules will present self peptides, viral peptides (synthesized by the cell itself) or peptides from microbes ingested in phagosomes. Class II MHC molecules, for their part, will present peptides from ingested microbes in vesicles (this type of molecule is only expressed in cells with phagocytic capacity). MHC molecules are only stably expressed on the cell membrane if they have a charged peptide: the presence of the peptide stabilizes the structure of the MHC molecules, the "empty" are degraded inside the cell. Peptide-loaded MHC molecules can remain on the membrane for days, long enough to ensure that a suitable T cell recognizes the complex and initiates an immune response.
In each individual, MHC molecules can present both foreign peptides (from pathogens) and peptides from the individual's own proteins. This implies that, at any given time, only a small proportion of the MHC molecules in a cell will present a foreign peptide: most of the peptides it presents will be its own, since they are more abundant. However, T lymphocytes are capable of detecting a peptide presented by only 0.1%-1% of MHC molecules to initiate an immune response.
Self peptides, on the other hand, cannot initiate an immune response (except in autoimmune diseases), because T cells specific for self antigens are killed or inactivated in the thymus. However, the presence of self peptides associated with MHC molecules is essential for the supervisory function of T lymphocytes: these cells are constantly patrolling the body, checking for the presence of self peptides associated with MHC molecules and triggering an immune response in the rare cases in which they detect a foreign peptide.
MHC molecules in transplant rejection
MCH molecules were identified and named precisely for their role in transplant rejection between mice of different inbred strains. In humans, the MHC molecules are the leukocyte antigens (HLA). It took more than 20 years to understand the physiological role of MHC molecules in presenting peptides to T cells.
As noted above, each human cell expresses 6 MHC class I alleles (one HLA-A, -B, and -C allele from each parent) and 6-8 MHC class 2 alleles (one HLA- DP and -DQ, and one or two HLA-DR from each parent, and some combinations of these). MHC gene polymorphism is very high: it is estimated that there are at least 350 HLA-A alleles in the population, 620 HLA-B alleles, 400 DR alleles, and 90 DQ alleles. Since these alleles can be inherited and expressed in many different combinations, each individual will probably express some molecules that will be different from the molecules of another individual, except in the case of identical twins. All MHC molecules can be targets for transplant rejection, although HLA-C and HLA-DP have low polymorphism, and are probably of minor importance in rejection.
In the case of a transplant (organ or stem cell), the HLA molecules function as antigens: they can trigger an immune response in the recipient, causing rejection of the transplant. The recognition of MHC antigens in cells of another individual is one of the most intense immune responses known. The reason why individuals react against another individual's MHC molecules is fairly well understood.
During the maturation process of T lymphocytes, they are selected based on the ability of their TCR to weakly recognize "self peptide:self MHC" complexes. For this reason, in principle, T lymphocytes should not react to a "foreign peptide:foreign MHC" complex, which is what will appear in the transplanted cells. However, it appears that what is occurring is a type of cross-reaction: the T cells of the recipient individual can make a mistake, because the donor's MHC molecule is similar to their own in the TCR-binding region (the variable region of the MHC is located in the zone of binding to the peptide that they present). For this reason, the recipient individual's lymphocytes interpret the complex present in the cells of the transplanted organ as "foreign peptide:self MHC" and they trigger an immune response against the "invading" organ, because they perceive it in the same way as their own infected or tumor tissue, but with a much higher number of complexes capable of initiating a response. The recognition of the foreign MHC molecule as their own by T lymphocytes is called allorecognition.
Two types of transplant rejection mediated by MHC molecules or human leukocyte antigens (HLA) can occur:
- hyperagudo rejection: occurs when the receiving individual presents preformed anti-HLA antibodies, before the transplant; this may be due to the prior realization of blood transfusions (which includes lymphocytes of the donor, with HLA molecules), to the generation of anti-HLA during pregnancy (against the HLA of the father present in the fetus) and by the realization of a previous transplant;
- acute humoral rejection and chronic dysfunction of the transplanted organ: it is due to the formation of anti-HLA antibodies in the receptor, against HLA molecules present in the endothelial cells of the transplant.
In both cases, an immune reaction is produced against the transplanted organ, which can generate lesions in it, which leads to loss of function, immediate in the first case and progressive in the second.
For this reason, it is essential to perform a cross-reaction test between donor cells and recipient serum, to detect the presence of preformed anti-HLA antibodies in the recipient against the donor's HLA molecules, and avoid hyperacute rejection. Normally, HLA-A, -B, and -DR molecules are matched: as the number of mismatches increases, 5-year post-transplant survival decreases. Full matching only exists between identical twins, but there are currently worldwide donor databases that allow optimizing the HLA match between a potential donor and a recipient.
Exogamy and CMH
In mice, sheep, fish (like the stickleback), birds and humans, there are studies that would indicate that mate selection has to do with the dissimilarity in the CMH and its relationship with the smell of the eventual mate. There is some evidence, especially in mice and sticklebacks, and less so in humans, that the scent of individuals with different HSCs is preferred, favoring diversity and thus a better immune system of offspring.
In humans, mate choice is determined by multiple factors, such as facial symmetry, psychological aspects, skin condition, and perhaps body odor. The results of the studies have shown some relationship between mate preferences and MHC dissimilarity. This would bring as a consequence a greater genetic variability of the descendants.
Contenido relacionado
Psilurus incurvus
Obesity
Bougainvillea



