Lead(II) nitrate
The chemical compound lead(II) nitrate is an inorganic salt of lead and nitric acid. It is a colorless crystal or white powder, and a very stable and strong oxidant. Unlike other lead(II) salts, it is soluble in water. Its main use, since the Middle Ages (under the name plumbum dulce) has been as a raw material in the production of numerous pigments. Since the 20th century, it has been used as a heat stabilizer for nylon and polyesters, and as a coating for photothermal imaging films. Its commercial production did not begin in Europe until the 19th century—in the United States until 1943—through a typical production process, using metallic lead or lead(II) oxide in nitric acid.
Lead(II) nitrate is toxic and probably carcinogenic. Therefore, it must be handled and stored under appropriate safety conditions.
History
Since the Middle Ages, lead(II) nitrate has been produced on a small scale as a base material for the production of coloring pigments, such as chrome yellow (lead(II) chromate) or chrome orange (lead(II) hydroxide chromate). lead(II)), or other similar lead compounds. In the 15th century, the German alchemist Andreas Libavius synthesized this compound, giving it the names plumb dulcis and calx plumb dulcis. Although the production process is chemically simple, production was minimal until the 19th century century, and no non-European production is known before the XX.
Chemistry
When lead nitrate is heated, it decomposes into lead(II) oxide, accompanied by a crackling noise (called decrepitation), according to the following reaction:
- 2 Pb(NO)3)2(s) → 2 PbO(s) + 4 NO2(g) + O2(g)
This property is what makes lead nitrate sometimes used in pyrotechnics (and more particularly, fireworks).
Chemistry in aqueous solution
Lead(II) nitrate dissolves in water to give a clear, colorless solution. This solution reacts with soluble iodides, such as potassium iodide (KI), producing a precipitate of lead(II) iodide. II) light yellowish-orange color. This reaction is often given as an example to demonstrate the chemical reaction of precipitation, due to the color change that is observed.
- Pb(NO3)2(aq) + 2KI(aq) → PbI2(s) + 2 KNO3(aq)
Other than lead(II) nitrate, lead(II) acetate is the only water-soluble lead compound. The other lead compounds are insoluble in water, not even the chlorides and sulfates (which are usually soluble) such as lead(II) chloride and lead(II) sulfate. This highlights the importance of lead(II) nitrate in the production of insoluble lead compounds by double decomposition reactions.
When 1 M sodium hydroxide solution is added to 0.1 M lead nitrate, basic nitrates are formed, even past the equivalence point. Up to halfway there, Pb(NO3)2 Pb(OH)2 is the predominant element, and once past, Pb(NO3)2 5Pb(OH)2 is formed. Surprisingly, no simple Pb(OH)2 is formed until pH 12 is reached.
Crystal Structure
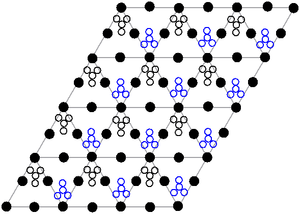
The crystal structure of solid lead(II) nitrate is determined by neutron diffraction. The compound crystallizes in a cubic system, with the lead atoms in a face-centered cubic system. Its space group is Pa3 (Bravais notation), and the length of each side of the cube is 784 picometers.
In the image, black dots represent lead atoms, white dots represent nitrate groups 27 pm above the plane of the lead atoms, and blue dots represent nitrate groups the same distance below of that same plane. In this configuration, each Pb atom is bonded to 12 oxygen atoms (bond length: 281 pm). All N-O bonds are identical (125 pm).
The academic interest in the crystal structure of this compound lies in part in the possibility of a free internal rotation of the nitrate groups in the crystal lattice at elevated temperatures, which, however, cannot be realized.
Complexation
Lead nitrate has interesting supramolecular chemistry due to the coordination of nitrogen and oxygen atoms. This interest is basically academic, but it has quite a few potential applications. Thus, the combination of lead(II) nitrate with pentaethylene glycol in a solution of acetonitrile and methanol, followed by slow evaporation, produces a new crystalline material, [Pb(NO3) 2(EO5)]. The crystal structure of this compound shows that the PEO chain is coiled around the lead ion in an equatorial plane, similar to a crown ether. The two bidentate nitrate ligands are placed in the trans configuration. The total coordination number is 10.
The complex formed by lead(II) nitrate, lead(II) perchlorate, and an N-dative bidentate bithiazole ligand is binuclear: in this complex, a nitrate group forms a "bridge" between lead atoms with a coordination number of 5 and 6. An interesting aspect of this type of complex is the presence of a physical void in the coordination sphere (i.e., the ligands are not arranged symmetrically around the ion). metal), which is probably due to the lone pair of electrons in lead. The same phenomenon is verified in lead compounds that have imidazole as ligand.
This type of chemistry is not always specific to lead nitrate; Other lead(II) compounds such as lead(II) bromide also form complexes, but nitrate is often used because of its solubility and bidentate nature.
Preparation
The compound is usually obtained by dissolving metallic or oxidized lead in an aqueous solution of nitric acid. Pb(NO3)2 anhydride can crystallize directly from the solution. There is no known industrial scale production.
- 3 Pb + 8 HNO3 → 3 Pb(NO)3)2 + 2 NO + 4H2O
- PbO + 2 HNO3 → Pb(NO)3)2 + H2O
Applications
Lead(II) nitrate has historically been used in the manufacture of matches and specialty explosives such as lead azide Pb(N3)2, in mordants and pigments (lead paints...), for coloring and printing fabrics, and in the production processes of lead compounds. Other more recent applications are, for example, as a heat stabilizer in nylon and polyesters, as a coating for photothermographic films, and in rodenticides.
Lead(II) nitrite is also a reliable source of pure dinitrogen tetroxide in the laboratory. When salt is carefully dried and heated in a steel container, it produces nitrogen(IV) oxide and oxygen. The gases are condensed and then fractionally distilled to give pure N2O4.
- 2 Pb(NO)3)2(s) → 2 PbO(s) + 4 NO2(g) + O2(g)
- 2 NO2 N2O4
Security
The hazards of lead(II) nitrate are those of soluble lead compounds in general, and to a lesser extent, those of the other inorganic nitrates. It is toxic, and its ingestion can lead to lead poisoning: symptoms include intestinal dysfunction, severe abdominal pain, loss of appetite, nausea, vomiting and cramps, and long exposures can lead to neurological and kidney problems. Lead compounds are known to be slow and cumulative poisons: more than 90% of absorbed lead is fixed in bone tissue, and is only eliminated over a long period of several years.
Children absorb lead from the GI tract more easily than adults, and are therefore at greater risk. Lead exposure during pregnancy has long been linked to high rates of spontaneous abortion, fetal malformations, and low birth weights. Given the cumulative nature of lead toxicity, children and pregnant women should not be exposed to soluble lead compounds, which is a legal requirement in many countries.
The International Center for Research on Cancer (CIRC) has classified inorganic lead compounds as potentially carcinogenic to humans (category 2A). They have been linked to kidney cancer and glioma in laboratory animals, and to kidney cancer, brain tumors, and lung cancer in humans, although studies of lead-exposed workers are often complex, as are arsenic.. A known function of lead is that of zinc substitute in numerous enzymes, such as δ-aminolevulinic acid dehydratase (or porphobilinogen-synthase) in the heme biosynthetic pathway and pyrimidine-5'-nucleotidase, important for the DNA metabolism.
Precautions should be taken before and during handling of lead(II) nitrate, for example by using protective equipment (safety glasses, rubber gloves...). Experiments with lead(II) nitrate should be carried out in fume hoods, and residues should not be released into the environment.
Contenido relacionado
Isomerism
Molybdenum
Serotonin