Hemoglobin A1
Hemoglobin A1 (HbA1) is a type of hemoglobin, also called adult hemoglobin or normal hemoglobin, which accounts for approximately 97 % of hemoglobin synthesized in the adult. It is made up of two alpha globins and two beta globins.
Structure and function
Hemoglobin A (HbA) is the most common adult form of hemoglobin and exists as a tetramer containing two alpha subunits and two beta subunits (α2β2). Each subunit contains a heme group to which heme molecules can bind. diatomic oxygen (O2). In addition to oxygen, subunit assembly and quaternary structure are known to play important roles in Hb affinity. When hemoglobin binds to O2 (oxyhemoglobin), it will bind to iron II (Fe2+) in heme and it is this iron ion that can bind and unbind oxygen for transport throughout the body. All subunits must be present for hemoglobin to pick up and release oxygen under normal conditions.
Summary
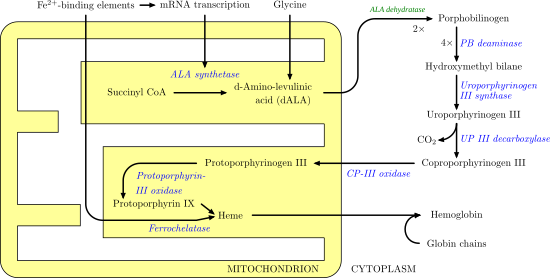
Heme synthesis
Heme synthesis involves a series of enzymatic steps that take place within the mitochondria and cytosol of the cell. First, in the mitochondria, condensation of succinyl CoA and glycine by ALA synthase occurs to produce 5-aminolevulinic acid (ALA). ALA then passes into the cytosol and after a series of reactions creates coproporphyrinogen III. This molecule moves back to the mitochondria where it reacts with protoporphyrin-III oxidase to produce protoporphyrin IX. Iron is enzymatically inserted into protoporphyrin via ferrochelatase to produce heme.
Globin synthesis
Globin synthesis takes place on ribosomes found within the cytosol. Two globin chains that have heme groups combine to form hemoglobin. One of the strings is an alpha string and the other is a non-alpha string. The nature of the non-alpha chain in hemoglobin molecules varies due to different variables. Fetuses have a non-alpha chain called gamma and after birth it is called beta. The beta chain will pair with the alpha chain. It is the combination of two alpha and non-alpha chains that creates a hemoglobin molecule. Two alpha chains and two gamma chains make up fetal hemoglobin or hemoglobin F (HbF). After the first five to six months after birth, the combination of two alpha chains and two beta chains form adult hemoglobin (HbA). The genes that code for alpha chains are located on chromosome 16, while the genes that code for non-alpha chains are located on chromosome 11.
Clinical significance
Due to the many steps and processes during the synthesis of hemoglobin, there are many places where errors can occur. Multiple enzymes are involved in the synthesis of hemoglobin and when these enzymes are deficient or do not function correctly, consequences such as mutations or deletions in the genes that encode the globin chain can occur. This results in disorders of the globin genes (hemoglobinopathies) which may be abnormal variants of the globin chain (sickle cell anemia) or reduced synthesis of the chain in erythroid cells (thalassemia) during the cellular process of hematopoiesis. These hemoglobinopathies are often inherited as autosomal recessive traits.
Alpha-Thalassemia
Alpha thalassemia (α-thalassemia) is defined by lack of production of the α-globin chain in hemoglobin, and is considered to carry a mutation affecting the α-globin chain on a single chromosome have "silent" thalassemia; of α, whereas if the mutation is in both, it is considered an α-thalassemia trait. α-thalassemia is found mainly in subtropical and tropical areas, where individuals carrying the gene make up 80–90% of the population. Like other hemoglobin-related disorders (sickle cell disease and β-thalassemia), it is hypothesized that α-thalassemia is selected within populations because carriers are better protected against falciparum malaria. Most α-thalassemia carriers are asymptomatic and are diagnosed if found after routine blood tests or before birth screening tests. Carriers of the single α-globin gene generally do not have profound fatigue or anemia because have a compensatory increase in the number of microcytic red blood cells. In contrast, mild carriers of α-thalassemia may have symptoms of anemia due to other factors not specifically related to the disorder: poor diet, drop in hemoglobin levels due to blood loss, or other illnesses.
The more severe form of α-thalassemia is a condition that begins in childhood in which there is no expression of the α-genes and results in a large production of Bart's hemoglobin.The most common cause of Hb Bart's is the inheritance of a deletion allele in which the functional genes for α-globin are missing from both parents. Hb Bart's is a tetramer of four gamma-globulin subunits and is ineffective in transporting oxygen to tissues due to its very high affinity for oxygen. This usually results in fatal hydrops fetalis and associated symptoms. they include intrauterine anemia, decreased brain growth, edema, skeletal deformities, and cardiovascular deformities that could lead to heart failure.
Beta-thalassemia
Beta-thalassemia (β-thalassemia) is an inherited mutation of the β-hemoglobin gene that causes reduced synthesis of the β-hemoglobin chain. Most mutations are point mutations that affect transduction, transcriptional control and splicing of the hemoglobin β gene and gene product. Individuals with one gene mutation (heterozygosity) are considered to have β-thalassemia minor (carrier or β-thalassemia trait), while those with two gene mutations (homozygosity or compound heterozygosity) are diagnosed with β-thalassemia or intermediate.
Due to the lack of beta-globin, accumulation of alpha-globin subunits and alpha tetramers begin to accumulate, causing red blood cell damage. People of Asian, Middle Eastern, and Mediterranean descent have a much higher incidence of β-thalassemia. Wide variation in disease phenotypes and genotypes has been determined because more than 200 different thalassemia-associated mutations have been found in the beta-globin gene. Individuals with β-thalassemia major usually require medical attention within the first 2 years of life and require regular blood transfusions to survive. Patients presenting with the disorder later generally do not require transfusions and are diagnosed with thalassemia intermedia.

]
Sickle Cell Disease
HbS, sickle cell hemoglobin, is the most common variant of hemoglobin and arises due to an amino acid substitution in the b-globin subunit at the sixth glutamic acid residue to valine. There are different forms of sickle cell anemia. HB SS which is the most common and severe form of sickle cell anemia. Hb SC is due to the inheritance of Hb S from one parent and Hb C (hemoglobin C) from the other parent. Hb S beta thalassemia is the least common and is experienced in patients who have inherited hemoglobin beta thalassemia from one parent and HbS from the other. In addition, there is sickle cell trait (HbAS) which is defined by having both HbA and HbS.. This makes the individual heterozygous for sickle cell. It is estimated that there are about 300 million individuals with sickle cell trait in the world population, and that about 100 million of these are found in sub-Saharan Africa. There is also a higher prevalence of sickle cell trait in areas where malaria is commonly found, with the prevalence in some parts of Africa and Saudi Arabia being up to 25% and 60%, respectively. Individuals who have HbAS have around 40% HbS, 56% HBA, and are generally asymptomatic unless there is severe lack of oxygen to the body (hypoxia) which can lead to symptoms of sickle cell disease. However, HbAS does not cause vaso-occlusive crises, which are known to be associated with sickle cell disease.
Patients who are homozygous for HbS have multifilament fibers that induce a change in the shape of red blood cells from biconcave disks to elongated crescents. Sickle formation is reversible after reoxygenation of hemoglobin, therefore red blood cells can go through sickle and normal cycles depending on the concentration of oxygen present in the blood stream. The sickle-shaped red blood cells lack flexibility and stick to the walls of blood vessels, slowing or stopping the flow of oxygen to nearby tissues. This decrease in oxygen to the tissues causes a vaso-occlusive crisis that presents as muscle pain and tissue damage. Some of the symptoms of sickle cell disease include fever, fatigue from anemia, swelling of the hands and feet, stroke, and organ failure. Current treatments include blood transfusions that help increase the number of normal red blood cells, bone marrow transplants to help the patient's body make healthy red blood cells, and medications to help alleviate the symptoms mentioned above.
Reference values
- Male: 13-17 g/dL
- Female: 12-15 g/dL
- Child: 11-16 g/dL
- Newborn: 14-24 g/dL
- Infant: 10-15 g/dL
Contenido relacionado
Raven (disambiguation)
Dna viruses
Pathological types of breast cancer