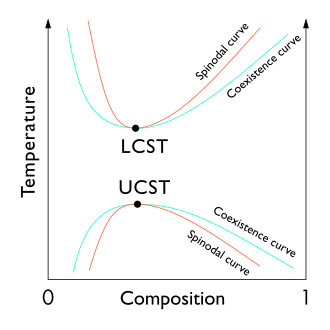Critical point (thermodynamics)
In thermodynamics and physical chemistry, a critical point is that limit for which the volume of a liquid is equal to that of an equal mass of vapor or, in other words, at which the liquid and vapor densities are equal. If the densities of liquid and vapor are measured as a function of temperature and the results are plotted, the critical temperature can be determined from the point of intersection of both curves.
Mathematical conditions of the critical point
At the critical point it is verified that:
(▪ ▪ P▪ ▪ v)T=Tc=0{displaystyle left({partial P; over partial v;}right)_{T=T_{c}}=0}
(▪ ▪ 2P▪ ▪ v2)T=Tc=0{displaystyle left({partial ^{2}P; over partial v^{2};}right)_{T_{c}}=0}
being P{displaystyle P} Pressure, v{displaystyle v} the molar volume, T{displaystyle T} temperature and Tc{displaystyle T_{c}} the critical temperature of the system considered.
Liquid–vapour critical point
Introduction
For simplicity and clarity, the generic notion of critical point is best introduced by discussing a specific example, the vapor-liquid critical point. This was the first hotspot to be discovered and is still the best known and studied.
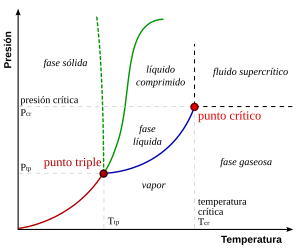
The figure on the right shows the schematic PT diagram of a pure substance (as opposed to mixtures, which have additional state variables and richer phase diagrams, discussed below). The commonly known solid, liquid, and vapor phases are separated by phase boundaries, that is, pressure-temperature combinations where two phases can coexist. At the triple point, all three phases can coexist. However, the liquid-vapor boundary ends at an endpoint at critical temperature Tc and critical pressure pc. This is the critical point.
In water, the critical point occurs at 647.096 K (373.946 °C) and 22.064 megapascals (217.75 atm).
In the vicinity of the critical point, the physical properties of the liquid and the vapor change drastically, and both phases become more and more similar. For example, liquid water under normal conditions is nearly incompressible, has a low coefficient of thermal expansion, has a high dielectric constant, and is an excellent solvent for electrolytes. Near the critical point, all of these properties turn into the exact opposite: water becomes compressible, expandable, a poor dielectric, a poor solvent for electrolytes, and prefers to mix with nonpolar gases and organic molecules.
At the critical point, there is only one phase. The heat of vaporization is zero. There is a stationary inflection point on the constant temperature line (critical isotherm) on a PV diagram. This means that at the critical point:
- (▪ ▪ p▪ ▪ V)T=0,{displaystyle left({frac {partial p}{partial V}}}{right)_{T}=0,}
- (▪ ▪ 2p▪ ▪ V2)T=0.{displaystyle left({frac {partial ^{2}p}{partial V^{2}}}}}}right)_{T}=0. !
Above the critical point there is a state of matter that is continuously connected (can transform without phase transition) to both the liquid and gaseous states. It is called supercritical fluid. The common textbook knowledge that any distinction between liquid and vapor disappears beyond the critical point has been challenged by Michael Fisher and Benjamin Widom, who identified a p-T line separating states with different asymptotic statistical properties (Fisher line -widom).
Sometimes the critical point is not manifest in most thermodynamic or mechanical properties, but is "hidden" and is revealed in the appearance of inhomogeneities in the elastic moduli, marked changes in the appearance and local properties of the disaffinated droplets, and a sudden increase in the concentration of defect pairs.
History
The existence of a critical point was first discovered by Charles Cagniard de la Tour in 1822 and named by Dmitri Mendeleev in 1860 and Thomas Andrews in 1869. Cagniard showed that CO2 could liquefy at 31 °C at a pressure of 73 atm, but not at a slightly higher temperature, even at pressures up to 3000 atm.
Theory
If the above expression is resolved (▪ ▪ p/▪ ▪ V)T=0{displaystyle (partial p/partial V)_{T}=0} for van der Waals equation, you can calculate the critical point as
- Tc=8a27Rb,Vc=3nb,pc=a27b2.{displaystyle T_{text{c}}={frac {8a}{27Rb},quad V_{text{c}}=3nb,quad p_{text{c}}}={frac {a}{27b^{2}}}}}} !
However, the van der Waals equation, based on mean field theory, is invalid near the critical point.
To analyze the properties of fluids in the vicinity of the critical point, reduced state variables relative to the critical properties are sometimes defined.
- Tr=TTc,pr=ppc,Vr=VRTc/pc.{displaystyle T_{text{r}}={frac {T}{T_{text{c}}}}}}}},quad p_{text{r}}={frac {p}{p}{p_{text{c}}}}},quad V_{text{r}}}{frac {V}{RT_{text{text{c}{c}{ !
The principle of corresponding states states that substances with equal pressures and reduced temperatures have equal reduced volumes. This relationship is approximately true for many substances, but is very imprecise for high values of pr.
For some gases, there is an additional correction factor, called Newton's correction, which is added to the critical temperature and critical pressure calculated in this way. These are empirically derived values and vary within the range of pressures of interest.
Mixtures: liquid-liquid critical point
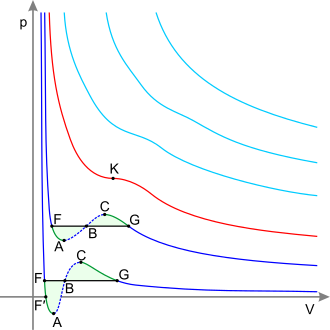
The liquid-liquid critical point of a solution, which occurs at the critical temperature of the solution, occurs at the boundary of the two-phase region of the phase diagram. In other words, it is the point at which an infinitesimal change in some thermodynamic variable (such as temperature or pressure) leads to the separation of the mixture into two distinct liquid phases, as shown in the polymer-solvent phase diagram to the right.. Two types of liquid-liquid critical points are the upper critical solution temperature (UCST), which is the hottest point at which cooling induces phase separation, and the lower critical solution temperature (LCST), which it is the coldest point at which the heating induces the phase. separation.
Mathematical definition
From a theoretical point of view, the liquid-liquid critical point represents the temperature-concentration extreme of the spinodal curve (as can be seen in the figure on the right). Therefore, the liquid-liquid critical point in a two-component system must satisfy two conditions: the spinodal curve condition (the second derivative of the free energy with respect to concentration must equal zero) and the condition of the extreme (the third derivative of free energy with respect to concentration must also equal zero or the derivative of spinodal temperature with respect to concentration must equal zero).